
DRAWBACKS OF THE QUANTUM MECHANICAL MODEL OF THE ATOM As a result, this model is based on probability rather than certainty. Quantum mechanical models of atoms use the complex shape of orbits (also known as electron clouds), which are the volumes of space in which electrons are likely to be present. QUANTUM MECHANICAL MODEL OF ATOM STRUCTURE 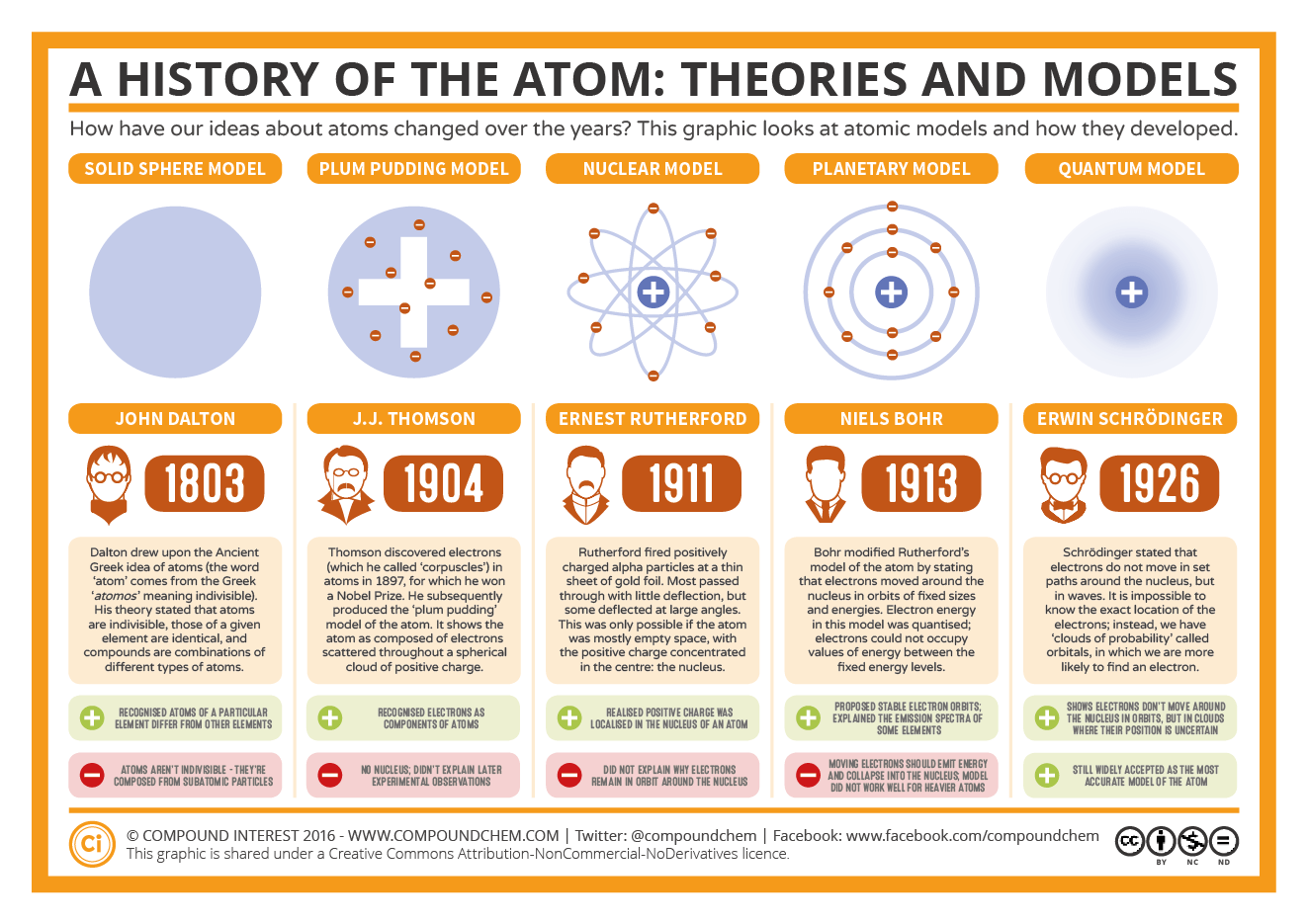
It also teaches us that no two electrons in the same system can be in the same energy state at the same time and that all energy states are filled from the lowest to the highest levels. Random density is always advantageous and is understood as | ψ | 2.Īccording to quantum theory, an electron can only exist in a limited number of quantized energy levels. The likelihood of an electron being placed on a positive web page within an atom is proportional to the square of the orbital wave characteristic at that location. The orbital wave properties of an atom contain all the statistics about the electrons in the atom, and quantum mechanics can extract these statistics. Since an electron can have many wave functions, it has multiple atomic orbitals.Įach wave’s characteristics or atomic orbitals have a specific shape and power. Electrons occupy atomic orbitals when their miles are characterized by wave characteristics. The wave characteristics (ψ) of electrons in an atom are called atomic orbitals. Where ψ represents the wave characteristics of the electron. Therefore, we can calculate the only danger of putting an electron in a particular function. 
The proven answer to the Schrodinger wave equation is the quantized power of the electron, which is the end result of the wave-like properties of the electron.Īccording to Heisenberg’s Uncertainty Principle, it is not possible to know the specific function and momentum of an electron. That is, it can have a positive performance value. FEATURES OF QUANTUM MECHANICAL MODEL OF ATOM Quantum mechanics models describe the possibility of placing electrons within an atom by describing the main energy level, orbital (arbitrary level), and spin. The quantum mechanical model of the atom was created by solving the Schrodinger equation. Schrodinger’s equation mathematically determined the regions that an electron was most likely to be found in the orbital lobes.In 1926, Schrodinger used the duality of electron wave particles to create and solve difficult mathematical formulas that accurately describe the behavior of electrons in a hydrogen atom. 
Schrodinger’s theory made use of electrons as waves, treating them as clouds of negative charge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
